Over the past few years, I worked with Sian Duss, a very skilled and talented PhD student in the lab of Johannes Bohacek, to dissect the role of noradrenaline release in the hippocampus. I’m very excited that the manuscript describing this work is now online as a preprint: Locus coeruleus noradrenaline elicits response profiles distinct from natural arousal in hippocampal neurons and astrocytes.
We used two-photon imaging of interneurons, astrocytes, and pyramidal cells in head-fixed mice, while optogenetically stimulating the primary source of noradrenaline, the locus coeruleus (LC). In addition, we used fiber photometry to calibrate our LC stimulation protocol and to compare optogenetically evoked noradrenaline release with that observed during behavioral paradigms.
For the main findings, please check out the manuscript itself. In this blog post, I’d like to highlight a few results that I personally find fascinating but that are less central in the paper.
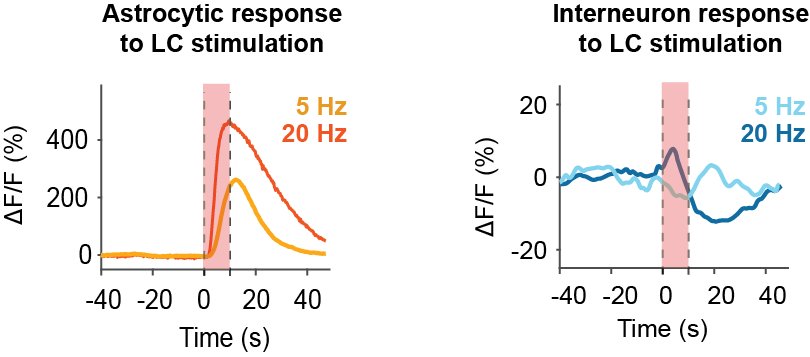
1. Astrocytes are much more sensitive to noradrenaline than neurons
Based on our two-photon calcium imaging data, pyramidal neurons and interneurons show little or no response to weaker LC stimulation and respond robustly only to strong stimulation. Astrocytes, in contrast, are highly sensitive and respond even to the lowest detectable increases in LC activity. This positions astrocytes ideally to mediate noradrenergic effects in hippocampal circuits. I find it great to have this very direct comparison across all three cell types – pyramidal cells, interneurons, and astrocytes – using the same experimental approach for each cell type.
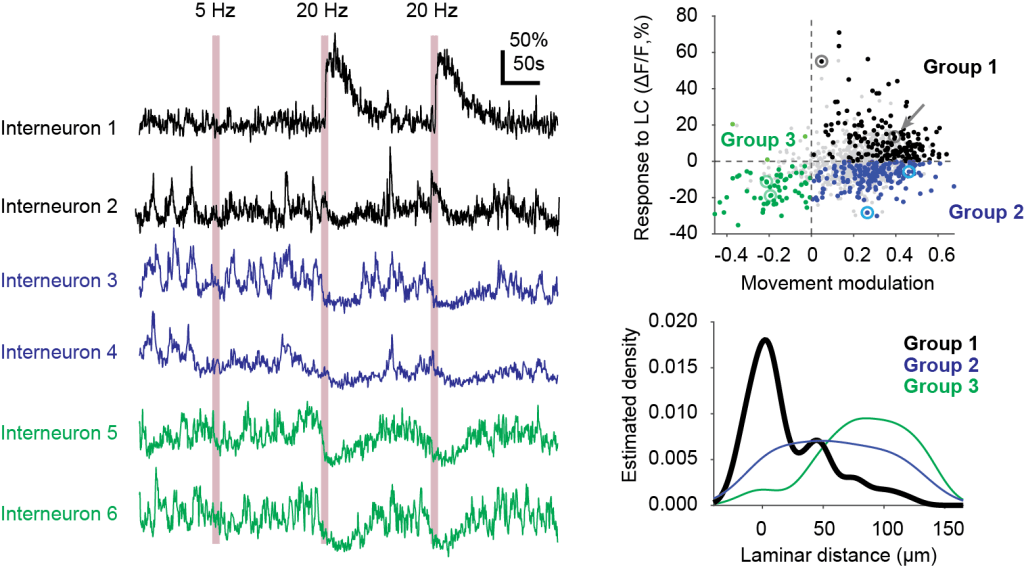
2. Interneurons responsive to noradrenaline are located close to the stratum radiatum
Previous studies have already described that specific functionally defined interneurons are located in specific laminae or sub-laminae of hippocampus (e.g., Geiller et al., 2020). Here, we found that interneurons responding to noradrenaline release upon optogenetic LC stimulation are preferentially located near the pyramidal cell layer, close to the stratum radiatum.
3. Not all astrocytes are equal
We found that some astrocytes are much more sensitive than others when responding to locus coeruleus activation. These differences were consistent across repeated stimulations and across days. This adds evidence, from a functional perspective, that astrocytes are not a homogeneous population, even within the same hippocampal region.
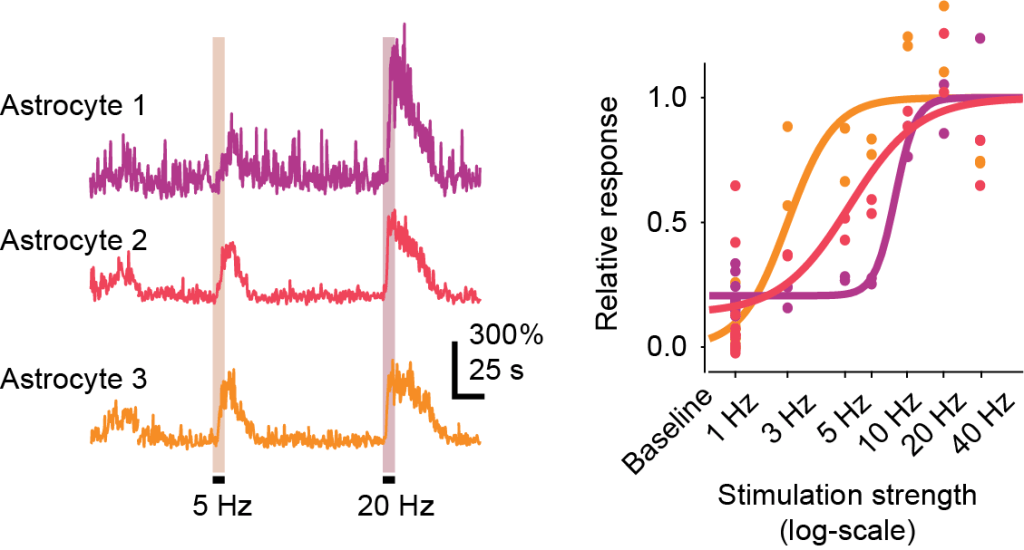
A functionally defined diversity of astrocytes. Some astrocytes respond at low noradrenaline levels (orange examples), others only at intermediate levels (red), again others only at high levels of noradrenaline release. Adapted from Figure 3 of Duss et al., 2026, under CC BY-NC 4.0 license.
For many more details on the cellular responses to locus coeruleus stimulation, and how these compare to response profiles during natural arousal, check out the preprint. Happy to hear thoughts or questions about the work!
Got any thoughts about not all astrocytes being equal? I’ve been more and more leaning towards the idea that variability between individual neuronal cells is understated in general for the past few years. I’ve not seen this explicitly tackled almost at all, and as for astrocytes I would expect something like this to be less measurable, so it’s interesting to see.
Interesting question! The idea that not all astrocytes are equal and fulfill different tasks in the brain has become a very popular idea during the last ~5 years among astrocyte researchers. I find the idea interesting but also somewhat puzzling.
It is known that astrocytes tile the brain in non-overlapping domains (at least in rodents). Given this organization, I wonder: if an individual astrocyte is specialized for a particular function, for example, a specific form of gliotransmission, why would that function be confined to just its own domain and not be more uniformly available across neighboring regions?
I’m still struggling to reconcile this apparent functional diversity of astrocytes with their uniform and non-overlapping coverage of space.
For the diversity of neurons, I can readily accept that it makes sense. Diversity of properties is more stable towards perturbations, offers more diverse computational resources, and is easy to establish. I don’t see why evolution (or development) should not favour a certain amount of neuronal diversity.
But I’m curious: from which perspective are you thinking about neuronal diversity? Functional imaging? Or something entirely different like transcriptomics?